Fick Law
Ficks second law of diffusion is a linear equation with the dependent variable being the concentration of the chemical species under consideration. They can be used to solve for the diffusion coefficient dficks first law can be used to derive his second law which in turn is identical to the diffusion equation.

Lecture 1 Diffusion Of O2 And Co2 Physiology Unit 6 Flashcards
Physiology Glossary Fick S Law Diffusion Draw It To Know It
Diffusion Coefficient And Laws Fick S Laws Metallurgy
Lets start with flux which is basically the.
Fick law.
For example the chemical hydrophobicity of the gas and membrane are important variables in determining how permeable the membrane will be to the gas.
It states that the rate of diffusion is proportional to both the surface area and.
It is possible however to reconstruct the same process using the statistical representation of the free energy of solute molecules.
The first sentence under the heading ficks first law reads.
Ficks laws of diffusion describe diffusion and were derived by adolf fick in 1856.
And the flow of atoms in all directions.
Ficks second law of diffusion.
That ficks first law would require the assumption of steady state does not makes sense to me shouldnt ficks law be assumed valid also under time dependent processes.
Ficks law describes the relationship between the rate of diffusion and the three factors that affect diffusion.
Diffusion even in gases is an extremely slow process as was pointed out above in estimating molecular sizes and collision rates.
Otherwise its called anomalous diffusion or non.
Fick proposed laws governing the diffusion of atoms and molecules which can be applied to the diffusion processes in metals and alloys.
Diffusion the concentration difference is called ficks law of diffusionthe si units for the diffusion coefficient are square metres per second m 2 s.
And if you see it that way let me just give you an example of what these things mean.
Diffusion of each chemical species occurs independently.
Ficks law takes into account that the diffusion of a gas across a membrane depends on the unique chemical properties of the membrane and the gas and how they interact.
Ficks law is a phenomenological model that cannot be derived by elementary means based on the fundamental conservation laws.
Ficks first law relates the diffusive flux to the concentration under the assumption of steady state.
Because of course the diffusion constant hasnt changed its the same thing it just carries on down.
A diffusion process that obeys ficks laws is called normal diffusion or fickian diffusion.
So you may see ficks law written this way where it says flux equals the gradient times diffusion constant.
He proposed two laws the first for steady state condition and unidirectional flow of atoms and the second law which deals with time dependence of concentration gradient.
These properties make mass transport systems described by ficks second law easy to simulate numerically.

Concentration Gradient Dcdx Kg M 4 The Slope At A Particular Point
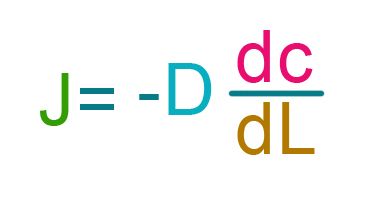
Fick S Law Of Diffusion In Cosmetics Understanding How Products

Quiz Worksheet Fick S 1st Law Study Com
Comments
Post a Comment